|
Across a period, effective nuclear charge increases as electron shielding remains constant. In general, atomic radius decreases across a period and increases down a group. What is the atomic radius trend on the periodic table? As you can see from the diagrams, the same atom could be found to have a different radius depending on what was around it. The radius of an atom can only be found by measuring the distance between the nuclei of two touching atoms, and then halving that distance. How do you find the radius of an element? Which of the following is correct order of increasing atomic size? The effect of the greater number of principal energy levels outweighs the increase in nuclear charge and so atomic radius increases down a group.ģ) The correct order of radii is: The atomic size decreases across a period from left hand side to right hand side. Group Trend Higher principal energy levels consist of orbitals which are larger in size than the orbitals from lower energy levels. Li, Na, K, Rb, Cs: (A) Li < Na < K < Rb < Cs. 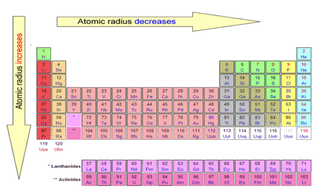
What is the increasing order of atomic radius? 
Thus, helium is the smallest element, and francium is the largest. As can be seen in the figures below, the atomic radius increases from top to bottom in a group, and decreases from left to right across a period. How do you rank elements in order of increasing atomic radius?Ītomic radii vary in a predictable way across the periodic table.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
